July 1, 2024
Radionetics provides Lilly access to proprietary GPCR targeting small molecule radiopharmaceuticals. Radionetics receives $140 million upfront payment and Lilly obtains the exclusive right to acquire Radionetics for $1 billion.
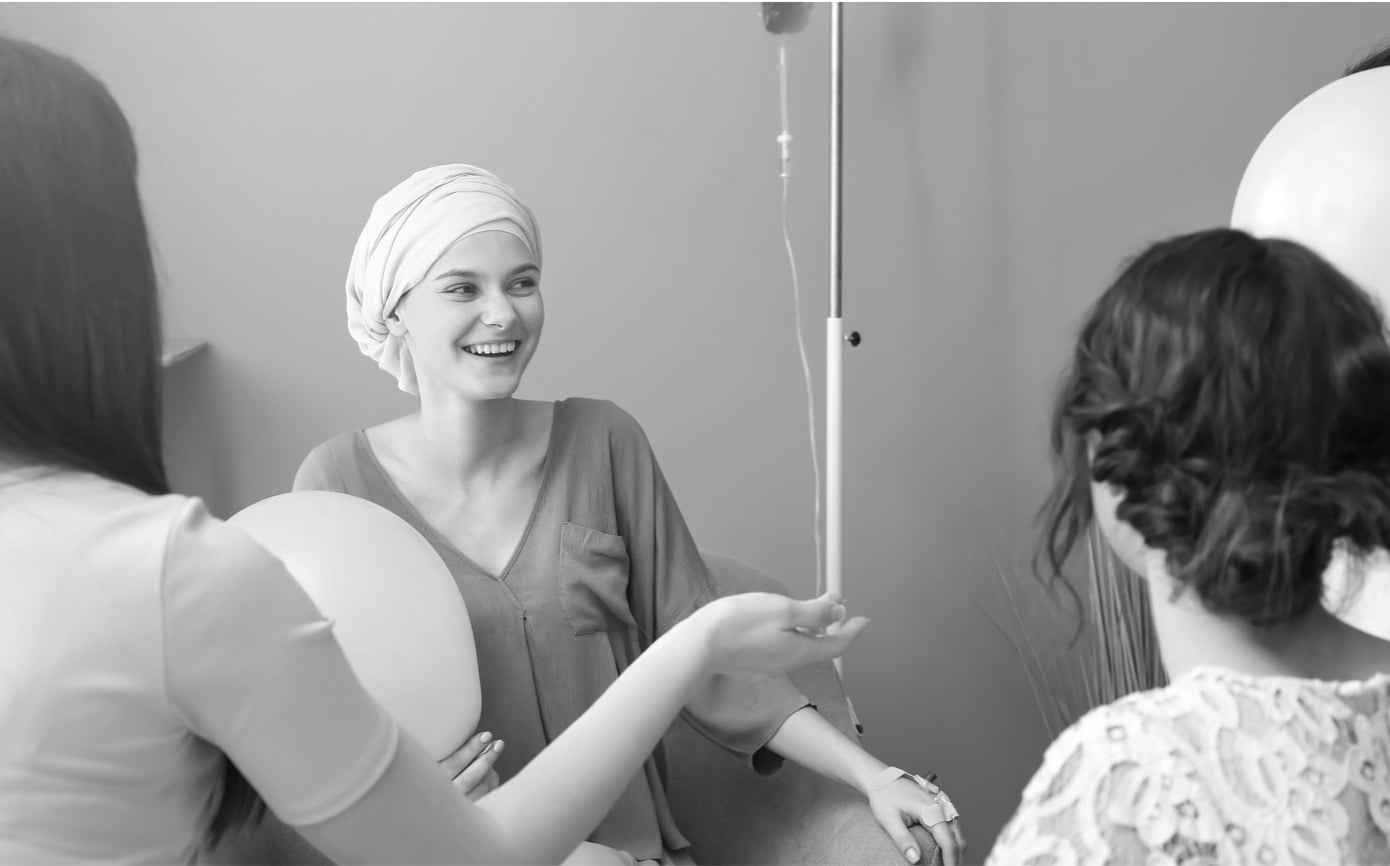
Part of a treatment strategy known as theranostics, radiopharmaceuticals can be used for both therapy and diagnosis.
Radiopharmaceuticals, when tethering a radionuclide suitable for imaging, create a map of cancerous tumors in the body that provides precise diagnostic information. From the imaging results, the patients who are most likely to respond are identified and treated with individualized regiments of the therapeutic version of the same radiopharmaceutical. This precision medicine approach seeks to make substantial improvements in clinical outcomes.

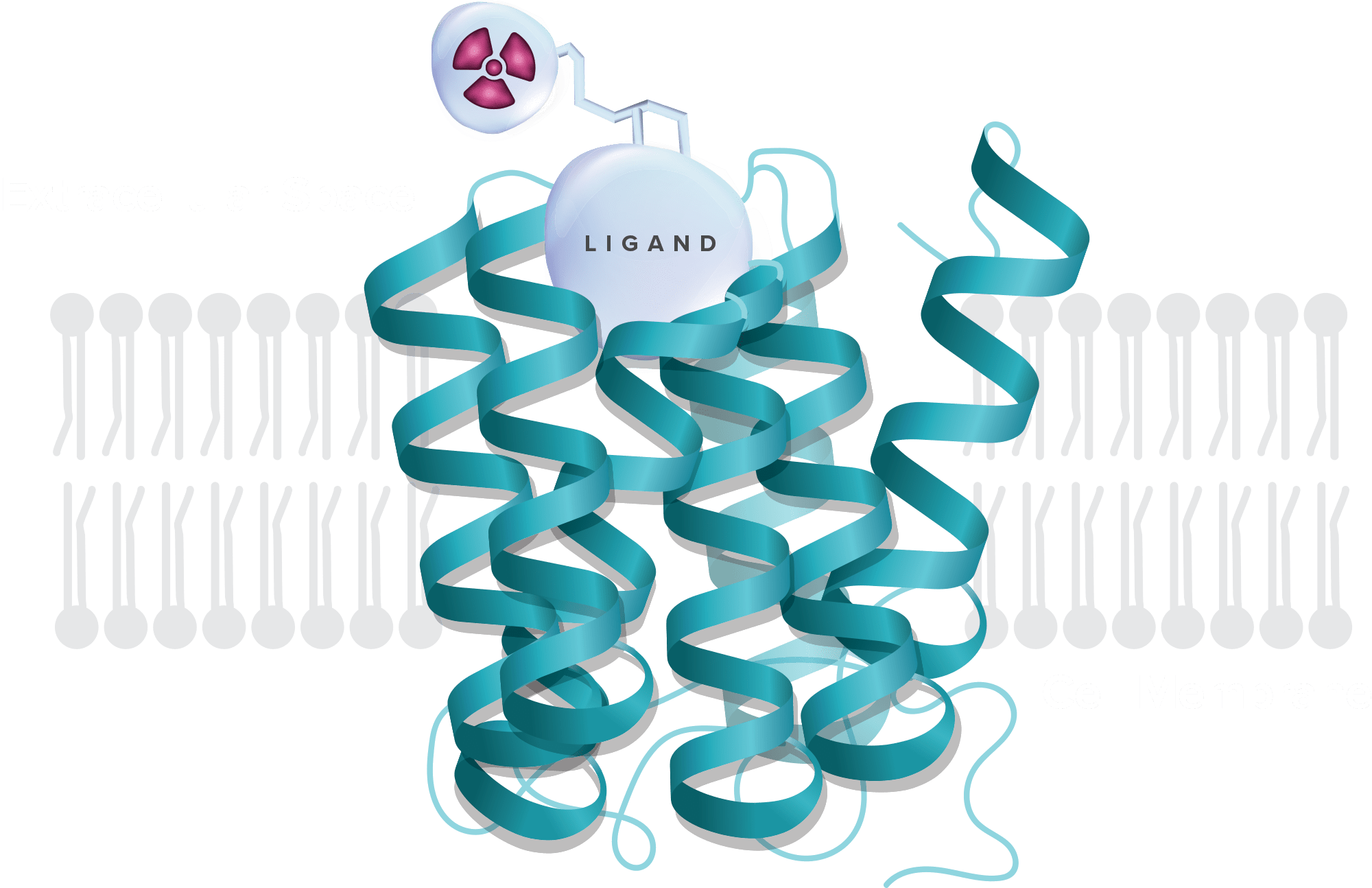
Small molecules, tethered with powerful radioisotopes, are designed to exquisitely bind to receptors overexpressed in cancer and deliver powerful radiation to kill tumors, without affecting normal tissue.
Our unique approach marries a vast knowledge of GPCR biology with unrivaled proficiency in small molecule medicinal chemistry to develop potent and highly selective radiopharmaceuticals. We take a fit-for-purpose approach, pinpointing the precise target and selecting the ideal small molecule ligand paired with the optimal radioisotope to achieve a therapeutic effect.
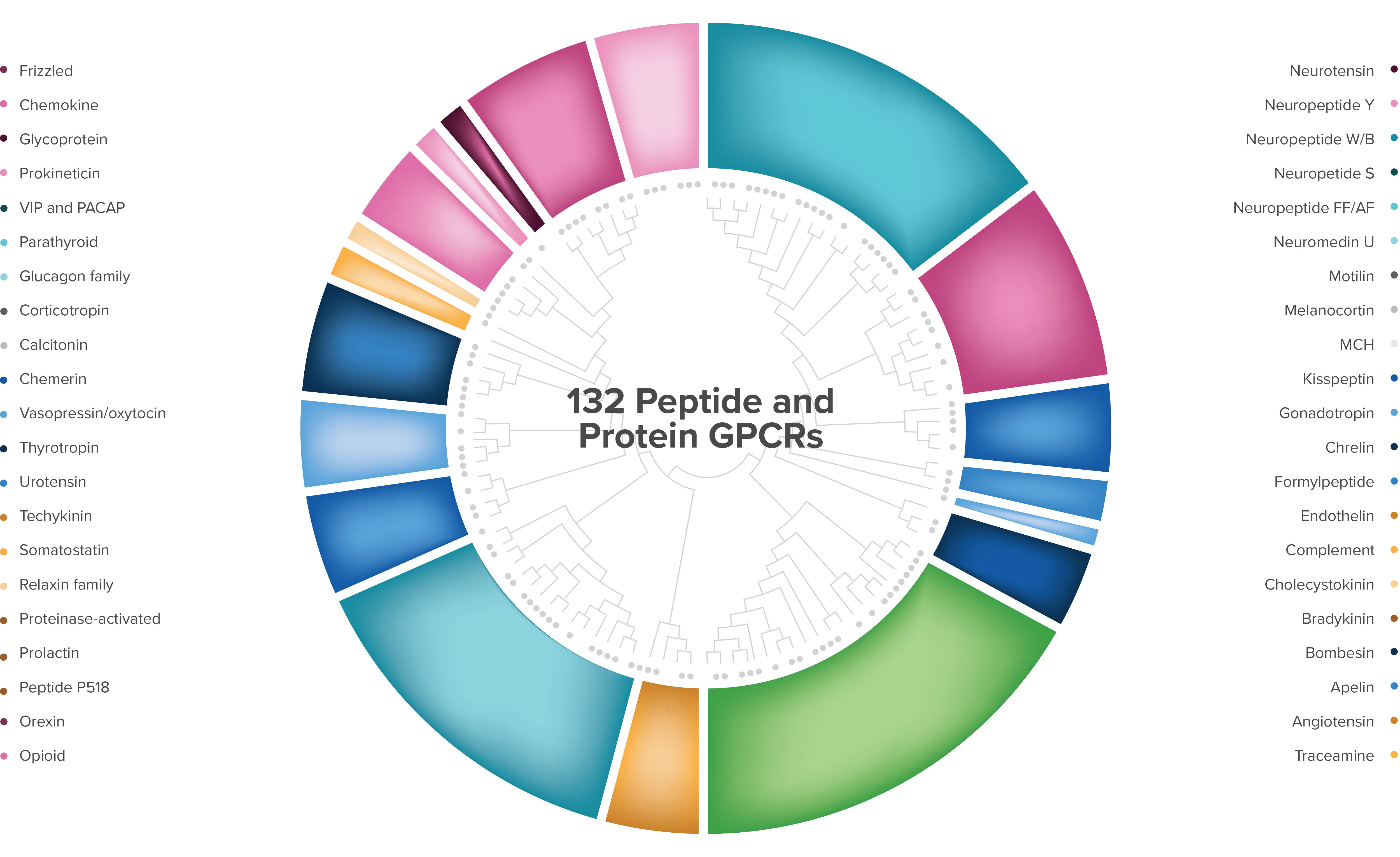
Largest gene family in humans
132 known protein/receptor pairs
>65 with increased tumor expression
Radionetics is advancing a pipeline of novel small molecule radioligands targeting GPCRs for the treatment of a broad range of cancers, including breast cancer, lung cancer, and other indications of high unmet need.












Our team is highly collaborative and dynamic, and we are doing exciting and novel research driving radiotherapeutics into the clinic and towards patients.
Radionetics is headquartered in San Diego, California, in the heart of the thriving life sciences community. Our team includes talented individuals who are experts in their respective fields and united by a shared passion for improving the lives of cancer patients.
Radionetics has a competitive total compensation package that includes bonus opportunity; equity; medical, dental, vision, life/AD&D, short-term, and long-term disability insurance; 401(k) retirement plan; 4 weeks of paid time off annually; and generous paid holidays.
We invite you to review our open positions and submit your application if there is a suitable match with your background and experience.

Umesh Gangadharmath, Ph.D. is Senior Vice President, Technical Operations and has focused his career on radiopharmaceutical development and manufacturing in both academic and commercial settings. He has over 16 years of experience in researching and developing novel radiopharmaceuticals in the areas of oncology, cardiology and neurology for use in positron emission tomography (PET) imaging and has established GMP radiopharmaceutical manufacturing sites globally to support clinical trials. Dr. Gangadharmath’s breadth of expertise spans from preclinical development, support of CMC sections for IND submissions, early clinical research, through full clinical production. Most recently, he was Chief Operating Officer at Optimal Tracers, a CDMO with the responsibility of supporting radiopharmaceutical production for biopharmaceutical companies. He spent the last 10 years building Optimal Tracers from concept to a mature, fully functioning GMP radiopharmaceutical manufacturing facility. Dr. Gangadharmath and his team at Optimal Tracers were also involved in establishing a fully GMP radiopharmaceutical manufacturing facility for clinical research at University Hospitals Cleveland Medical Center. Dr. Gangadharmath has held previous roles as Director for the biomedical cyclotron facility at UCLA and as Senior Scientist, Radiochemistry at Siemens Molecular Imaging Biomarker Research. He was part of the team that invented TAUVID during his work at Siemens Molecular Imaging Biomarker Research (Flortaucipir; Avid Radiopharmaceuticals, Inc., a wholly owned subsidiary of Eli Lilly and Company) for imaging tau pathology and is well-published in the field of radiopharmaceuticals. He also has served as a member of United States Pharmacopeia (USP) Radioactive Drugs Expert Panel and played a key role in setting up a molecular imaging program at Loma Linda University Medical Center. He received his M.Sc. and Ph.D. in Inorganic Chemistry from Karnatak University Dharwad, India and completed a postdoctoral fellowship at UCLA under the supervision of Dr. Hartmuth Kolb.